The British Society of Sexual Medicine (BSSM) published its first formal position statement on enclomiphene in February 2026 -- and for an unlicensed therapy that has lived in regulatory limbo for over a decade, it is the closest thing to mainstream society endorsement enclomiphene has ever received.
The statement, authored by Foster, Choo, Patel, Kirby, and Hackett in the World Journal of Men's Health, stops short of recommending routine use. But it explicitly identifies enclomiphene as a legitimate option for selected men with secondary hypogonadism -- particularly those who want to preserve fertility or cannot tolerate injectable or topical testosterone.
Key Takeaways
- BSSM published its first enclomiphene position statement in February 2026
- Endorses use only by experienced clinicians in specialist or research settings
- Targets men with secondary hypogonadism who want fertility preservation or are intolerant of standard TRT formats
- Confirms enclomiphene remains unlicensed in the UK and unapproved by the FDA
- Calls evidence "limited" and warns against routine use outside specialist clinics
- Does not change U.S. regulatory status -- compounding pharmacies remain the only legal source
What the BSSM Position Statement Actually Says
The statement is short and deliberately narrow. It does not declare enclomiphene a first-line therapy. It does not recommend it for general practice. What it does is far more practical: it draws a clinical lane in which experienced clinicians can prescribe enclomiphene without operating outside society guidance.
The BSSM defines that lane around four criteria:
- Secondary hypogonadism only. The patient must have low testosterone driven by reduced pituitary signalling -- low or low-normal LH and FSH -- not primary testicular failure.
- Fertility preservation as a primary goal. This is the strongest indication. Conventional TRT suppresses spermatogenesis within weeks; enclomiphene maintains it.
- Specialist setting. The statement explicitly limits use to "experienced clinicians within specialist or research settings." General practice is not the intended venue.
- Patient counselling on unlicensed status. Patients must be told the drug is not licensed in the UK and not approved by the FDA, and must consent to that.
The society also flagged what enclomiphene is not a candidate for: primary hypogonadism, men with normal LH and FSH despite low testosterone, and routine off-label substitution for standard TRT in men without fertility goals.
Why This Matters Now
For over a decade, enclomiphene has occupied an awkward space. The pharmacology is clean, the human safety record is reasonable, and the fertility-preservation case is uncontested. But the FDA rejected the new drug application in 2015 over manufacturing and clinical-data concerns, and no sponsor has resubmitted since.
That left enclomiphene as a compound-only drug in the U.S. and an unlicensed import in most of Europe. Mainstream urology and endocrinology societies have largely declined to take a position, leaving prescribers to make individual judgments without society backing.
The BSSM statement breaks that silence. It is the first major Western society to formally describe a clinical lane for the drug.
What Changed in the Evidence Base
The position statement leans on a small but consistent body of evidence:
- Wiehle et al. (2014). Phase II randomized trial in men with secondary hypogonadism showed enclomiphene normalized testosterone levels while preserving sperm counts; topical testosterone normalized levels but suppressed sperm counts. (PMID: 25044085)
- Kim et al. (2016). Two Phase III trials in obese hypogonadal men confirmed the same pattern. Enclomiphene raised testosterone, raised LH and FSH, and kept sperm counts in the normal range. Topical testosterone suppressed all three. (PMID: 26496621)
- Real-world cohort data (2019-2024). Smaller observational studies in specialist clinics have reported similar testosterone elevations with low side-effect rates.
The Phase III trials are now nearly a decade old. The BSSM statement frames the evidence as "limited" precisely because no sponsor has run the long-term safety and fertility-outcome studies that would clear regulatory bars.
The Specialist-Only Recommendation
The "specialist or research settings" language is the single most important constraint in the document. The BSSM is not endorsing telehealth-style mass prescribing. It is endorsing thoughtful, lab-driven use by clinicians who understand the HPG axis and can monitor LH, FSH, estradiol, and sperm parameters over time.
That is a meaningful gap from how enclomiphene is often marketed in U.S. telehealth -- as a "natural" or "fertility-friendly" alternative offered to almost anyone who walks into the funnel. The BSSM position effectively challenges that approach without naming it.
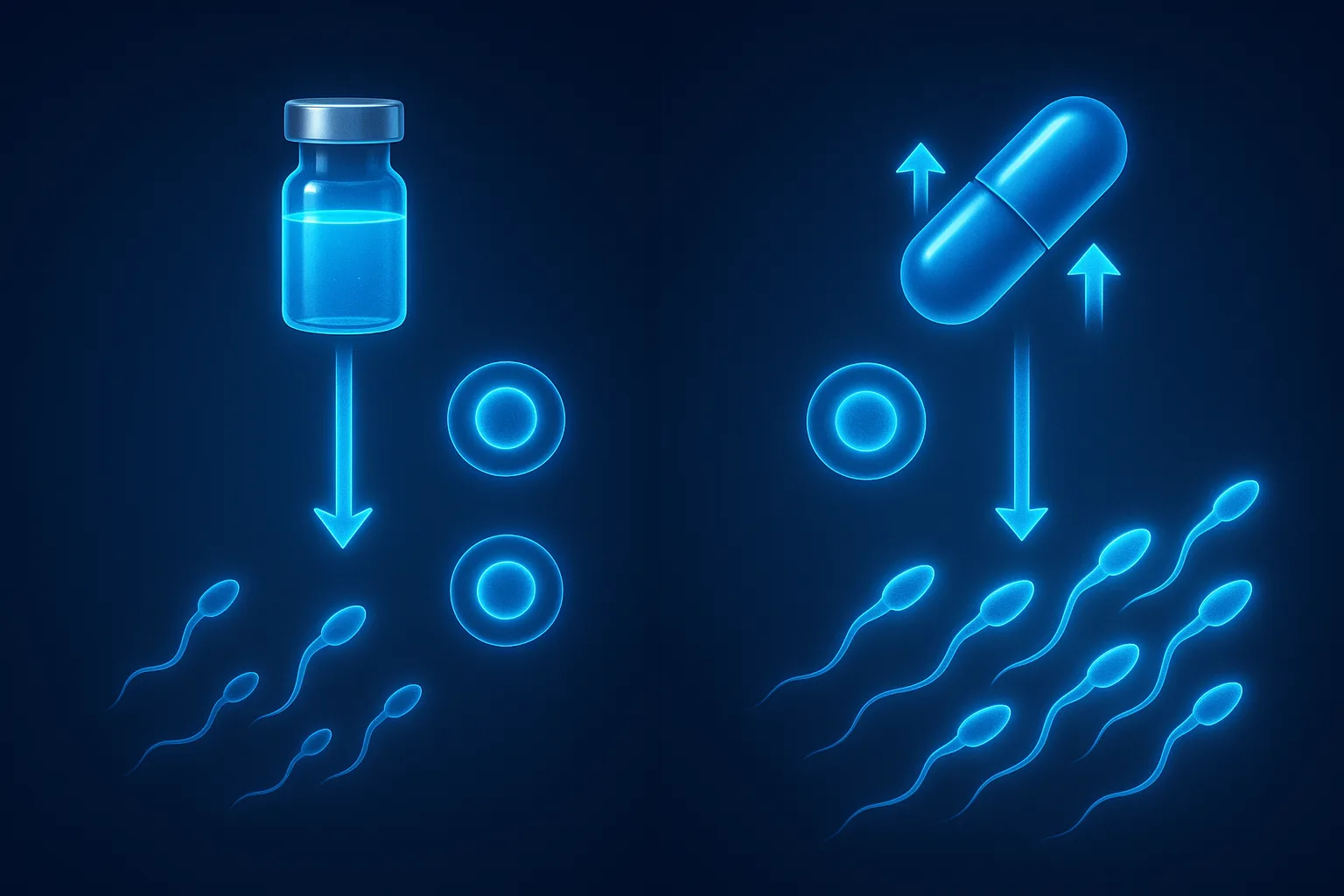
How Enclomiphene Differs from TRT in One Picture
The core mechanism difference is the entire reason enclomiphene exists as a separate therapeutic category.
| Variable | Enclomiphene | Standard TRT |
|---|---|---|
| Mechanism | SERM blocks estrogen feedback at hypothalamus/pituitary | Exogenous testosterone replaces endogenous production |
| LH / FSH | Increased | Suppressed (often near zero) |
| Spermatogenesis | Preserved | Suppressed within weeks |
| Testicular volume | Maintained | Typically shrinks 10-20% in year 1 |
| Route | Oral capsule, daily | Injection (typical), gel, pellet, oral |
| Stopping | Levels return to baseline within weeks | Variable HPG recovery, often months |
| Estradiol | May rise (managed by titration) | Variable, often manageable |
| Regulatory status | Compounded only in U.S., unlicensed in UK | FDA-approved, multiple licensed products |
The single most important row is spermatogenesis. For a man under 45 who wants children in the foreseeable future, that row alone often determines the choice -- and the BSSM statement validates that decision pathway.
For a deeper comparison of how to choose between these two approaches based on your specific labs and goals, read our enclomiphene vs TRT guide.
Who the BSSM Statement Targets
Reading the statement closely, four patient profiles fit cleanly:
1. Men in their 30s and early 40s with secondary hypogonadism
This is the prototypical candidate. Total testosterone under 300 ng/dL, low or low-normal LH and FSH, intact testes, no identifiable pituitary disease, and either active fertility goals or future fertility goals. Enclomiphene preserves the option of fathering children without adjunct medications.
2. Men who failed TRT due to side effects
A subset of men cannot tolerate injectable or topical testosterone -- skin reactions to gels, injection-site issues, supraphysiologic estradiol responses, or persistent hematocrit problems. The BSSM acknowledges this group as legitimate enclomiphene candidates.
3. Men coming off TRT who need HPG-axis restart
Although the BSSM does not specifically endorse "post-cycle" use, the underlying mechanism makes enclomiphene a logical tool for restoring endogenous production after exogenous testosterone has shut down the pituitary. This is the clinical scenario behind most off-label use today. Read our coming off TRT guide for how this fits a broader exit protocol.
4. Younger men who want to optimize testosterone without injections
Men in their 20s and 30s with mildly low or low-normal testosterone, intact fertility, and a strong preference for an oral therapy. The BSSM is more cautious about this group, framing it as a specialist judgment call rather than a clear indication.
Who the Statement Does Not Cover
The BSSM explicitly excludes routine use for:
- Men with primary hypogonadism (testes cannot respond to LH regardless of input)
- Men over 60 with established age-related decline (less response, longer trial needed)
- Men with elevated baseline LH/FSH
- Men with normal testosterone seeking "optimization" rather than treatment
- General-practice prescribing without specialist oversight