Key Takeaways: Menopausal hormone therapy started within roughly 10 years of the final menstrual period or before age 60 is associated with a 39% reduction in all-cause mortality, up to 50% reduction in coronary heart disease, and slower atherosclerosis progression. The same therapy started 10 or more years past menopause shows neutral or potentially harmful cardiovascular effects. The clinical principle, called the timing hypothesis or the window of opportunity, was confirmed by the ELITE trial, supported by re-analysis of the Women's Health Initiative, and is now embedded in major society guidelines. The FDA's 2026 removal of the broad black-box warning on HRT reflects this updated evidence. Practical translation: if you are in perimenopause or within several years of menopause and HRT is otherwise indicated, the time-sensitive case for starting is stronger than most women have been told.
The Question That Changed Menopause Medicine
For two decades, a generation of women was told that hormone replacement therapy was risky and probably unnecessary. That message was anchored to the 2002 Women's Health Initiative findings and the FDA black-box warning that followed. Prescriptions fell by more than 70%. Many primary care doctors stopped offering HRT. Many women who would have benefited went without it.
The Women's Health Initiative was a real trial with real findings. What was missing from the public translation was a critical detail: the average woman in the WHI was 63 years old at enrollment and more than a decade past menopause. The trial answered a specific question -- "what happens when we start HRT in older women, many of whom already have subclinical cardiovascular disease?" -- and the answer was, on average, not great.
It did not answer the more clinically relevant question: "what happens when we start HRT in a 50-year-old in perimenopause with vasomotor symptoms and no established vascular disease?" Subsequent re-analyses, the ELITE trial, the KEEPS trial, and a series of meta-analyses converged on a different answer to that question.
The framework that emerged is the timing hypothesis. It is now the dominant interpretation of the trial evidence and is embedded in 2026 guidelines from the Menopause Society, the British Menopause Society, the International Menopause Society, and -- as of late 2025 and into 2026 -- in updated FDA labeling.
What the Timing Hypothesis Actually Says
The clinical claim has three parts:
- Cardiovascular outcomes of menopausal hormone therapy depend on when it is started relative to vascular health, not on whether HRT is intrinsically "good" or "bad."
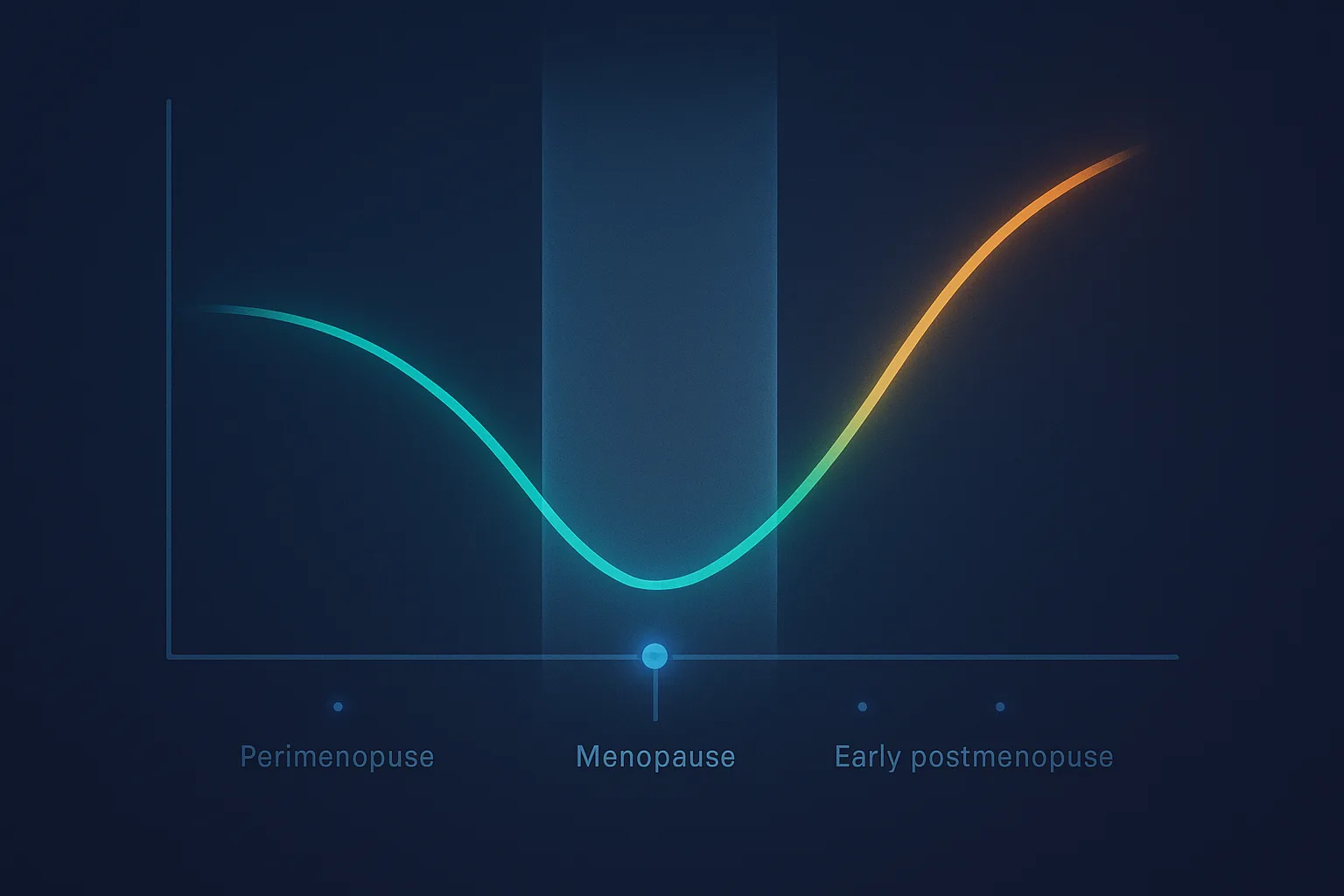
- Started early -- within roughly 10 years of menopause or before age 60 -- HRT slows atherosclerosis progression, reduces coronary heart disease, and reduces all-cause mortality.
- Started late -- more than 10 years past menopause, in women who already have subclinical atherosclerosis -- HRT does not reduce cardiovascular events and may destabilize existing arterial plaque.
The mechanism is now reasonably well understood. Estradiol acts on endothelium, vascular smooth muscle, and platelets in fundamentally different ways depending on the underlying state of the artery. In a vascularly young artery, estradiol promotes nitric oxide release, inhibits smooth muscle proliferation, and prevents the early steps of atherogenesis. In an artery with established lipid-rich plaque, the same hormone can increase plaque inflammation and contribute to plaque rupture and thrombus formation.
The 10-year mark is a population-level approximation. The actual variable is vascular age. Women with cardiometabolic risk factors -- diabetes, hypertension, dyslipidemia, smoking -- may exit the protective window earlier. Metabolically healthy women may have a slightly longer window. But for most clinical decisions, "within 10 years of menopause or before age 60" captures the relevant signal.
The Trial Evidence Stack
The timing hypothesis is not based on a single study. It rests on a coherent stack of randomized and observational evidence. Five datasets are central.
The Women's Health Initiative — Re-Analyzed
The WHI enrolled 27,347 postmenopausal women in two arms: conjugated equine estrogens alone (in women with prior hysterectomy) and CEE plus medroxyprogesterone acetate (in women with intact uteri). The 2002 stop announcement reported increased risk of breast cancer and cardiovascular events in the combined arm, which drove the practice change.
The 18-year cumulative follow-up published in 2017 produced a different headline. Across both arms, there was no significant difference in all-cause mortality between hormone therapy and placebo: 27.1% versus 27.6% [1]. When stratified by age at initiation, women who started HRT in their 50s showed lower coronary heart disease and all-cause mortality than the placebo group. Women who started in their 60s and 70s showed neutral or worse outcomes.
The same data, two readings: "HRT is risky" (2002, average-effect framing) versus "HRT effects depend on age at initiation" (2017, stratified framing). The second reading is the timing hypothesis.
The ELITE Trial
The Early versus Late Intervention Trial with Estradiol was designed specifically to test the timing hypothesis. ELITE randomized 643 healthy postmenopausal women -- stratified into early postmenopause (less than 6 years out) and late postmenopause (10 or more years out) -- to either oral 17β-estradiol or placebo. The primary endpoint was carotid intima-media thickness, a validated surrogate for atherosclerosis progression.
After median follow-up of 5 years, estradiol slowed CIMT progression in the early-postmenopause stratum but had no effect in the late-postmenopause stratum [2]. The interaction between time since menopause and treatment effect was statistically significant. ELITE is the cleanest randomized confirmation of the timing hypothesis.
KEEPS
The Kronos Early Estrogen Prevention Study enrolled 727 healthy women within 3 years of menopause and randomized them to oral conjugated equine estrogens, transdermal estradiol patch, or placebo, all with cyclical micronized progesterone. After 4 years, there was no difference in CIMT progression between groups [3]. KEEPS did not show benefit, but it also did not show harm, in a young postmenopausal population. Critically, KEEPS-Continuation follow-up suggested favorable long-term cognitive outcomes in the transdermal estradiol arm.
KEEPS is sometimes cited against the timing hypothesis, but the more accurate reading is that 4 years is a short window for atherosclerosis-progression endpoints in healthy women -- the trial may have been underpowered for the cardiovascular signal.
Salpeter Meta-Analyses
The 2004 and 2009 Salpeter meta-analyses pooled randomized HRT trials and stratified mortality outcomes by mean age at initiation. In the younger group (mean age 54), HRT was associated with a 39% reduction in total mortality [4]. In the older group (mean age 66), HRT showed no mortality benefit. The 2009 Bayesian re-analysis of 19 trials and 16,000 younger postmenopausal women confirmed a relative risk of 0.73 with high probability of net benefit.
These pooled estimates are consistent with the WHI age-stratified re-analysis and with ELITE. The convergence across designs strengthens the timing-hypothesis interpretation.
Hodis and Mack Synthesis
Howard Hodis and Wendy Mack -- ELITE's principal investigators -- published two synthesis papers in 2013 making the explicit case that the timing hypothesis represents a paradigm shift in primary cardiovascular prevention for women [5]. Their argument: HRT initiated in younger postmenopausal women has effect sizes on coronary heart disease prevention comparable to statins and antihypertensives, but with a different risk profile and indication footprint.
The broader synthesis paper extending this analysis through 2022 confirms that across observational and randomized data, HRT in women under 60 or within 10 years of menopause is associated with significant mortality and cardiovascular benefit [6].
What This Means in 2026
Three practical changes follow from the timing-hypothesis framework, all of which are already showing up in 2026 prescribing practice.
The FDA Black-Box Warning Is Being Removed
In late 2025 and into 2026, HHS and the FDA initiated removal of the broad black-box warning that had been on menopausal hormone therapy products since shortly after the WHI announcement. The agency cited the accumulated evidence that the warning -- which was applied uniformly across all HRT products and all patient ages -- substantially misrepresented the risk-benefit profile for women starting therapy near the menopausal transition.
The practical effect: many primary care providers and gynecologists who reflexively avoided HRT for the past 20 years are reconsidering. Women who were previously told HRT was too risky are being offered the therapy they should have been offered earlier. See HRT Black Box Warning Removed for the regulatory detail.
HRT Prescriptions Are Rising 72% Since 2021
Independent prescription data show menopausal hormone therapy prescriptions up 72% since 2021. The rise has been concentrated in women aged 40 to 55 -- exactly the population the timing hypothesis identifies as most likely to benefit. Telehealth platforms specializing in women's hormone care have driven much of this prescribing growth, in large part because they apply timing-hypothesis-aware protocols rather than the legacy avoid-HRT-by-default posture.
Guideline Convergence
Major society positions in 2026 align around timing-hypothesis-informed practice:
- The Menopause Society: HRT is appropriate first-line treatment for vasomotor symptoms in healthy women within 10 years of menopause and under age 60
- British Menopause Society: HRT benefit-risk profile is favorable for symptomatic women under 60 or within 10 years of menopause
- International Menopause Society: Timing of HRT initiation relative to menopause is a critical determinant of risk-benefit balance
- American College of Obstetricians and Gynecologists: Hormone therapy should be considered for symptomatic women without contraindications, with the most favorable profile in those starting within 10 years of menopause
The doctrinal convergence is striking after two decades of fragmented messaging.