
The only reason you can get testosterone prescribed through a video call right now is a temporary waiver that expires December 31, 2026. The DEA has renewed this waiver four times since 2020, but there's no permanent rule in place -- and the proposed replacement would require in-person visits that would effectively end telehealth TRT as it exists today.
If you're currently getting TRT through an online clinic, or considering starting, you need to understand what the rules actually are, which states already restrict access, and what's likely to happen next.
Key Takeaways
- The DEA's telemedicine waiver lets providers prescribe testosterone via video without an in-person visit -- but it expires December 31, 2026
- Without a permanent rule, the Ryan Haight Act would require in-person evaluation before any TRT prescription via telehealth
- 16 states already restrict or prohibit online TRT prescribing due to state-level regulations
- The FDA's potential descheduling of testosterone from Schedule III could permanently solve the telehealth access problem
- If you rely on telehealth TRT, take specific steps now to protect your access
Why Telehealth TRT Exists: The COVID Waiver
Before March 2020, prescribing testosterone through a video call was essentially illegal under federal law.
The Ryan Haight Act
The Ryan Haight Online Pharmacy Consumer Protection Act of 2008 requires an in-person medical evaluation before any practitioner can prescribe a controlled substance via telemedicine. Since testosterone is classified as Schedule III, this law applied directly to TRT.
In practice, this meant online TRT clinics couldn't exist in their current form. Every patient would need to see a doctor in person before receiving a prescription -- defeating the primary advantage of telehealth.
The COVID Emergency Changed Everything
When the COVID-19 public health emergency was declared in March 2020, the DEA temporarily waived the Ryan Haight Act's in-person requirement. Suddenly, DEA-registered practitioners could prescribe Schedule II through V controlled medications via audio-video telemedicine without ever meeting the patient face-to-face.
This waiver created the modern telehealth TRT industry overnight. Clinics like those in our best online TRT clinic comparison built their entire business models on this flexibility.
The 2026 Extension: What It Actually Says
On December 31, 2025 -- literally the last possible day -- the DEA and HHS jointly issued a fourth extension of telemedicine flexibilities through December 31, 2026.
What's Currently Allowed
| Rule |
Details |
| Who can prescribe |
Any DEA-registered practitioner |
| Substances covered |
Schedule II through V controlled medications (testosterone is Schedule III) |
| Visit type required |
Audio-video telemedicine (no audio-only for initial TRT prescriptions) |
| In-person requirement |
Waived -- no in-person visit needed before prescribing |
| Prescription limits |
Standard prescribing rules apply (no additional telehealth-specific limits) |
| State compliance |
Federal waiver doesn't override state restrictions |
What It Doesn't Change
The extension doesn't override three critical requirements:
- Prescriptions must be for legitimate medical purposes. You still need documented low testosterone (typically below 300 ng/dL on two morning draws) and symptoms.
- Providers must be licensed. The prescribing practitioner must hold a valid DEA registration and state medical license.
- State laws still apply. If your state restricts telehealth prescribing of controlled substances, the federal waiver doesn't help.
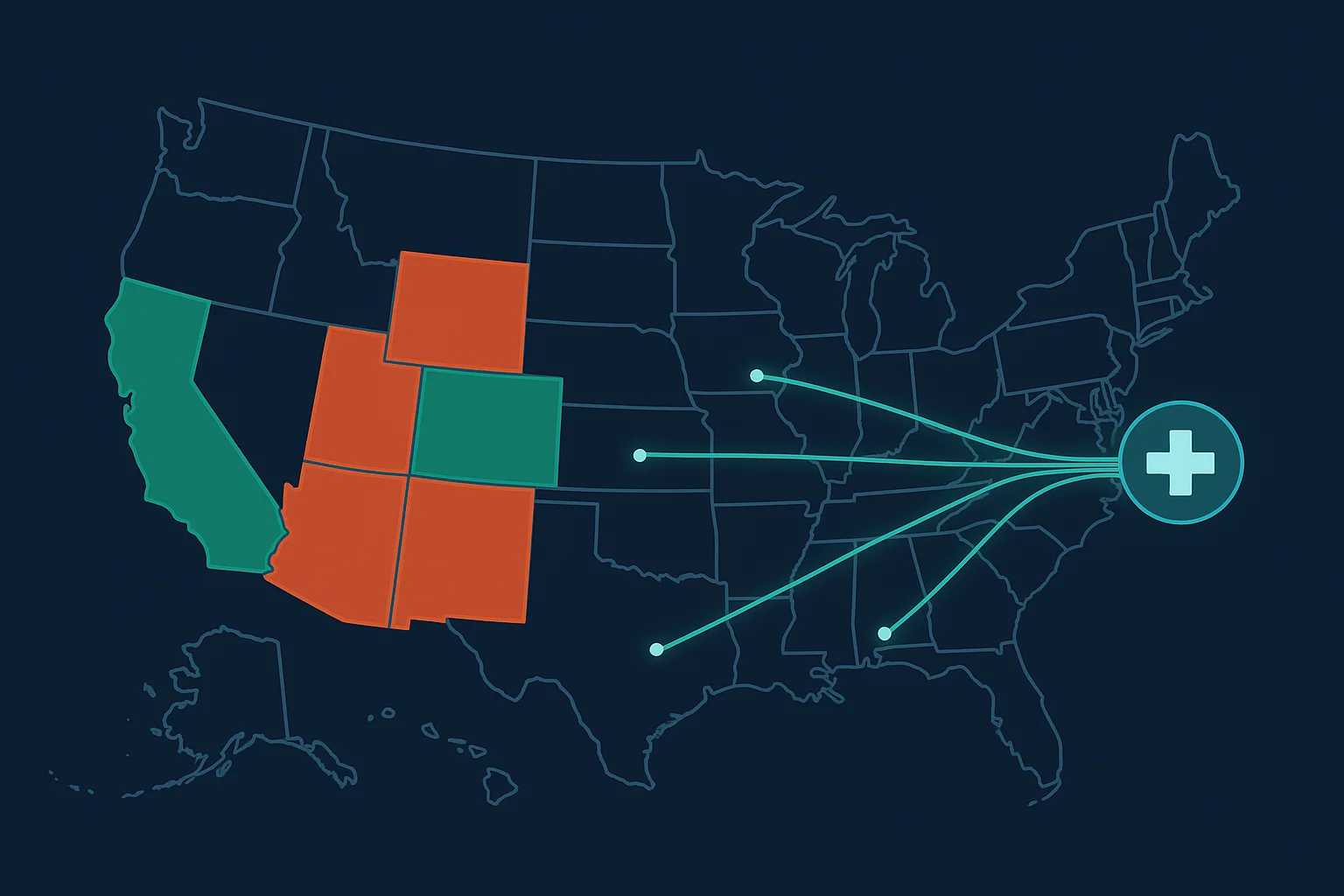
The 16 States That Restrict Telehealth TRT
Even with the federal waiver in place, state-level regulations create a patchwork of access. As of April 2026, approximately 16 states restrict or prohibit telehealth TRT prescribing:
| Restricted States |
|
| Alabama |
Mississippi |
| Arkansas |
Missouri |
| Connecticut |
North Carolina |
| Delaware |
North Dakota |
| Georgia |
Oklahoma |
| Hawaii |
Pennsylvania |
| Louisiana |
Rhode Island |
| Minnesota |
South Carolina |
These restrictions vary in severity. Some states require an initial in-person visit. Others prohibit telehealth prescribing of controlled substances entirely. A few restrict shipping of controlled substances into the state.
If you live in one of these states, your options include:
- Local clinics. Use our guide on how to choose a TRT clinic to find a provider near you.
- Hybrid models. Some clinics offer an initial in-person visit followed by telehealth management. See our online vs. local doctor comparison for a detailed breakdown.
- Travel for initial visit. A few telehealth clinics accept patients from restricted states if they complete one in-person visit at a partner location.
What Happens After December 2026
This is the question every telehealth TRT patient should be asking. There are three realistic scenarios.
Scenario 1: Another Extension (Most Likely)
The DEA has extended these flexibilities four consecutive years. The political cost of suddenly cutting off millions of patients' access to controlled substance prescriptions via telehealth is high. Another year-long extension is the path of least resistance.
Probability: High. This has happened every year since 2022.
Impact on your TRT: None. Business as usual for another year.
Scenario 2: Permanent Telehealth Rules (Best Case)
The DEA proposed a Special Registration for Telemedicine framework in January 2025 that would create permanent pathways for telehealth prescribing of controlled substances. However, this framework has not been finalized.
If permanent rules are adopted, they could include:
- A special DEA registration category for telehealth prescribers
- Standardized requirements for video visits and documentation
- Clear rules on prescribing across state lines
- Potentially more restrictive initial evaluation requirements than the current waiver
Probability: Moderate. The DEA has signaled intent but hasn't acted.
Impact on your TRT: Likely continued access with some new requirements.
Scenario 3: The "Telemedicine Cliff" (Worst Case)
If the waiver expires without extension or replacement, the Ryan Haight Act snaps back into full effect. Every patient receiving TRT via telehealth would need an in-person evaluation before their next prescription.
The proposed DEA rules (which were never finalized) would have required:
- An in-person evaluation before the initial prescription
- A second in-person visit within 30 days for prescriptions beyond a 30-day supply
- Dual record-keeping by both examining and prescribing physicians
- Precise timestamps on all visit documentation
As Defy Medical noted, the 30-day limitation is particularly problematic for TRT: steady testosterone levels typically aren't reached for 8+ weeks, meaning patients would need an in-person follow-up before the medication has even had a chance to work.
Probability: Low, but not zero.
Impact on your TRT: Significant disruption. You'd need to find a local provider or travel for in-person visits.
The Descheduling Wild Card
There's a fourth possibility that could make this entire issue irrelevant: removing testosterone from Schedule III.
In December 2025, an FDA expert panel recommended descheduling testosterone entirely. Their argument: testosterone doesn't meet the criteria for controlled substance classification -- a position the DEA and the American Medical Association have held since the original 1990 scheduling.
If testosterone is descheduled, the Ryan Haight Act no longer applies to it. Telehealth TRT prescribing would become permanently legal without any special waiver, the same way a doctor can prescribe blood pressure medication via telemedicine today.
FDA Commissioner Martin Makary has signaled openness to the panel's recommendations. The Endocrine Society and multiple medical organizations submitted formal comments supporting the change before the February 2026 deadline. But no timeline for a decision has been announced.
What to Do Now: 5 Steps to Protect Your Access
Whether you're currently on TRT or considering starting, take these steps:
1. Establish Care Before December 2026
If you're considering telehealth TRT, start now. Existing patient relationships may receive different treatment than new patients under any future rules. Most clinics in our best online TRT clinic guide can get you started within 1-2 weeks.
2. Keep Your Lab Records
Download and save every lab result. If you need to transition to a new provider -- whether due to regulatory changes or other reasons -- having a complete lab history avoids repeating expensive blood work. Our guide on how to read testosterone labs explains which values to track.
3. Know Your State's Rules
Don't assume the federal waiver covers you. Check whether your state has additional telehealth restrictions for controlled substances. The Center for Connected Health Policy maintains updated state-by-state information.
4. Have a Local Backup Plan
Identify a local urologist or endocrinologist who prescribes TRT. Even if you prefer telehealth, having a local provider relationship means you won't face a gap in treatment if the rules change. Our online vs. local doctor guide can help you evaluate local options.
5. Understand Your Clinic's Contingency Plan
Ask your telehealth provider directly: "What's your plan if the DEA waiver expires?" Established clinics should have answers. If they don't, that's a red flag.
The Bigger Picture
The telehealth TRT prescribing landscape is caught between two converging trends.
On one side, demand for online TRT has never been higher. An estimated 20 million American men have clinically low testosterone, and telehealth removed the biggest barrier to treatment: the hassle of finding a knowledgeable local provider, scheduling in-person appointments, and navigating a healthcare system that often dismisses men's hormonal concerns.
On the other side, regulators are trying to balance access with oversight. The DEA's concern -- preventing diversion and misuse of controlled substances -- is legitimate. But as the American Telemedicine Association has argued, testosterone has minimal diversion risk. It's non-addictive at therapeutic doses. Blood labs provide adequate clinical monitoring without in-person visits.
The most promising resolution is descheduling. If testosterone is removed from Schedule III -- as the FDA panel recommended and as medical organizations support -- the entire telehealth prescribing question becomes moot. Until then, the annual cycle of last-minute DEA extensions will likely continue.
References
- DEA. "DEA Extends Telemedicine Flexibilities to Ensure Continued Access to Care." Dec 31, 2025. DEA Press Release
- Federal Register. "Fourth Temporary Extension of COVID-19 Telemedicine Flexibilities for Prescription of Controlled Medications." Dec 31, 2025. Federal Register
- American Telemedicine Association. "The Imperative of Telemedicine Prescribing for Testosterone." ATA Blog
- CCHP. "State Telehealth Policies for Online Prescribing." CCHP
- Endocrine Society. "Society Submits Comments to FDA Expert Panel on Testosterone Replacement Therapy for Men." 2026. Endocrine Society


